TuberculosisThe burden of TB disease on human society is hard to overstate, as it claims more lives than any other infectious disease. The only vaccine in use is 100 years old. CD4 T cell immunity to TB is essential, but efforts to target these cells specifically have not yet delivered a successful vaccine candidate. The T-cell-centric paradigm in TB research risks becoming a conceptual hurdle to the adoption of other more holistic approaches.
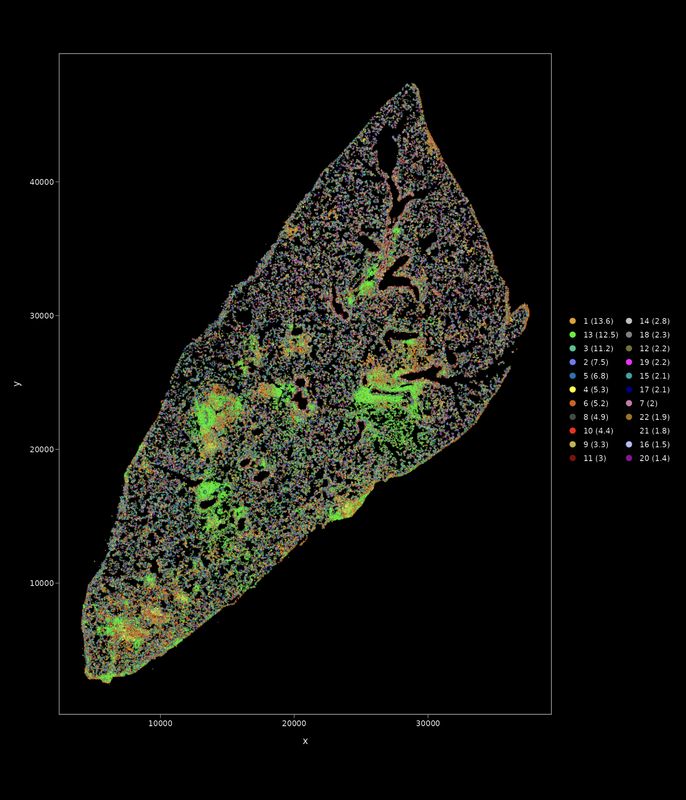
Our recent work suggests that CD4 T cells that interact with B cells in the lung have a better chance of providing long-lasting protection against pulmonary infection compared to more inflammatory T helper cells. Moreover, the accumulation of B cells in the lung, largely overlooked so far, correlates with reduced TB disease in humans and animal models of infection. B cells act as integral hubs with wide-ranging effects in the immune network, but their impact in TB hasn’t been adequately assessed. The adoption of a new, complementary perspective on TB from the point of view of antibodies and tissue resident B cells requires exhaustive foundational knowledge that is now technically within reach. By repurposing existing approaches (immunoproteomic antigen identification), pioneering new technology (single cell in situ transcriptomics), and linking multiple B cell features (specificity, transcriptome, B cell receptor repertoire, anatomic compartmentalization) with TB disease, we aim to establish this new paradigm. |
|
Influenza
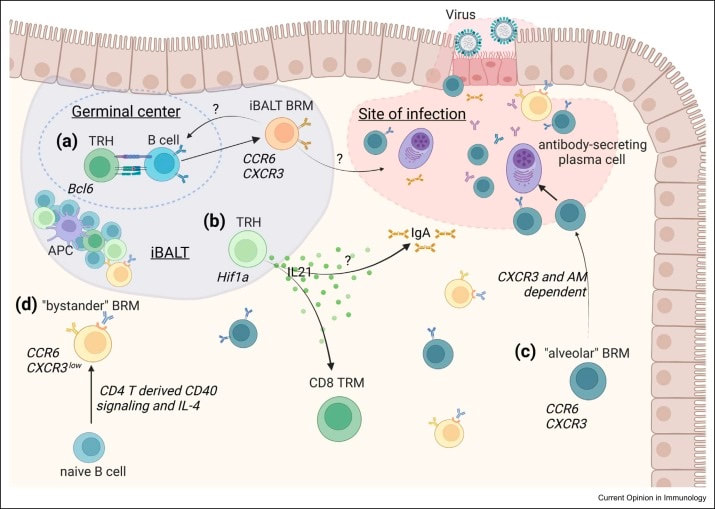
Influenza virus, a common airborne pathogen that infects the respiratory tract and undergoes continuous antigenic mutations, causes thousands of hospitalizations and deaths every year. For vaccines to be effective against influenza variants, we need a better understanding of how immune memory (both T and B lymphocytes) develops and persists in the lung mucosa. Sustained tissue resident memory can act as a sentinel of the immune response, rapidly responding to secondary infection challenge at the site of viral entry. |